Today, we will look at the major flaw of the German Panther tank, and look at the faulty metals used on German tanks during the Second World War which led to a large number suffering extreme cracking and spalling after receiving hit. This is supported by the metallurgical tests undertaken by the British, American, and Canadians during the war.
You’ve likely heard or subscribe to one of two ideas about German tank armor during the Second World War: either that it is terrible, or an excellent achievement of superior German engineering.
As always, its never quite that simple. So we’re here to help set the record straight, at least for the Panther.
Contents
Panther
The tank that would become the Panther started back in 1938 as a project for a new medium tank to replace the Panzer III and IV when the time came. This resulted in the VK 20 series, with designs being submitted by MAN, Krupp, and Daimler-Benz. This became a 20-ton tank that reached the prototype stage, but the project was stopped and the specifications changed to a 30-ton vehicle.
Work began on the new design in late 1941 between the German tanks design departments, with the aim to have prototypes ready by May 1942. Of the two proposals, it was the MAN design that was accepted. Work from this project resulted in the VK 30.02 (M), the first prototype Panther.
Read More The PaK 40 auf RSO – A Tank Destroyer with an Overly Large Gun
The vehicle itself looked good on paper; with a powerful anti-tank gun, well-sloped armor, and a good turn of speed, however it suffered initially from a lack of adequate testing, and a rush to get many in service for the Kursk offensive. As a result, they suffered heavily from mechanical issues and breakdowns often seen with vehicles that have not been adequately tested to root out flaws.

But rather than espouse the usual rhetoric, we will be looking at one of the more uncommon flaws of the Panther, one that also affected other German tanks, but none more so than this one, and that is the poor quality of the metal found in many examples.
These flaws would make the Panther somewhat of a liability, partially in combat, but more importantly in the aftermath where heavy damage received by the vehicle would make recovery and repairs much more difficult. This is especially bad when you are on the losing side, where the limited numbers of expensive tanks and the ability to recover and repair them in a timely manner is of the utmost importance.
So, let’s begin with German steel manufacturing during the war.
German Steel
Firstly, its important to understand what armor actually does. This is much, much more complicated than it seems, but in its most basic premise, armor absorbs and redistributes the energy imparted into it by an incoming projectile.
The projectile carries energy, if the armor has the appropriate properties to handle that energy, the projectile will not penetrate the steel. If the projectile carries more energy than the armor can handle, it will penetrate the steel. In practice, there are a huge amount of variables to consider which affect the outcome, and the situation quickly becomes extraordinarily complex.
Armor can be designed and created to better handle the energy from a projectile. Primarily, this is done by the steel simply absorbing the impact throughout its volume – hence why thicker armor plates provide better protection (in theory, at least). But armor can also be designed to interfere with the projectile itself to reduce its effectiveness; for example, shattering it and spreading out the concentration of energy.

Angling the armor can also help, as it effectively increases the thickness of the plate, and can cause a shell to be deflected or shatter.
The two primary types used in the construction of tanks by the Germans were Rolled Homogeneous Armour (RHA), and Surfaced Hardened variants of it. RHA is formed by hot rolling the steel to homogenize (organize into a uniform arrangement) the grain structure of the steel. This process increases the density and elongates the grain structure in the steel to form long lines, which help to spread the stress of an impact over a wider area.
RHA can be further heat-treated to form a hard outer layer, known as Surface of Face Hardening. Face hardened RHA is known as heterogeneous armor – heterogeneous meaning it contains different qualities (soft, impact absorbent interior, with a hard exterior).
Read More Object 770 – The 60 Ton Soviet Monster
Germany’s primary producers of the steel used during the Second World War were Krupp, United Steelworks, and others, each of whom had to send regular samples early on for inspection by the Supreme Command of Land Forces. This armor was split into seven categories based on thickness; from 5 – 14.5 mm plate, and then varying in thickness up to 200 mm plate at the extreme end.

The intermediary plates of armor in the 35-50 mm and 50-80 mm range are the most relevant to this discussion, as those involved surface treating to form heterogeneous armor, which would give the material both strong and ductile qualities. This was not something common in steel production, but by altering the crystal matrix to have varying, non-linear grain it could be achieved, albeit slowly.
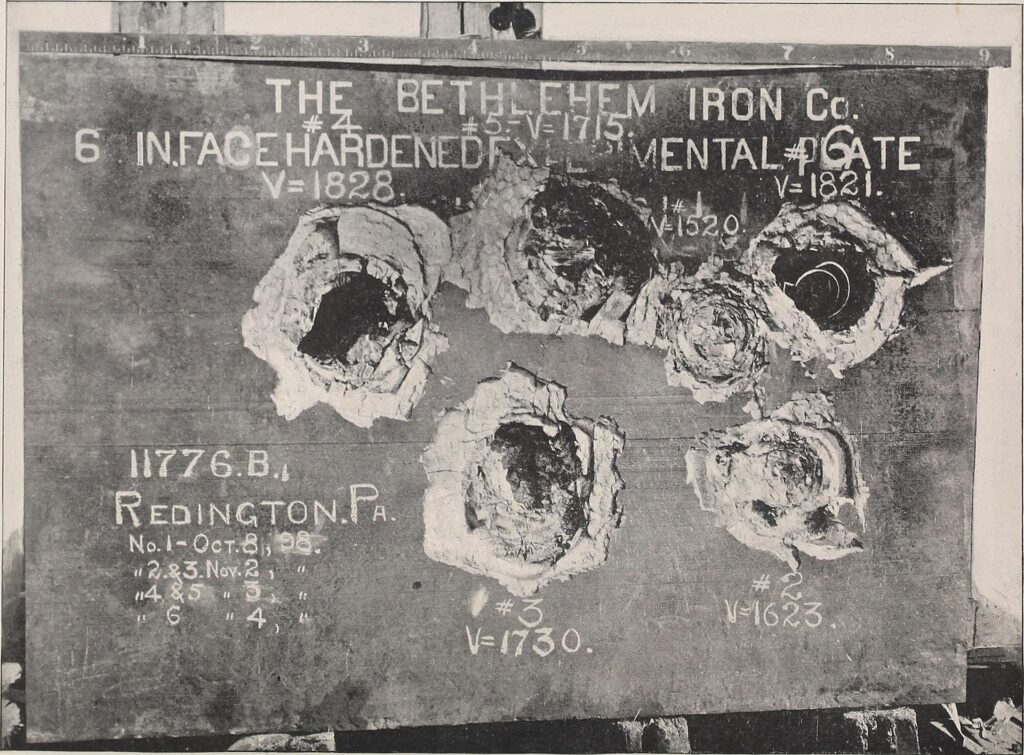
This surface-hardened armor was used notably on Panther, Tiger, and Tiger II through the war up until late 1944.
To create this heterogeneous armor, Germany used flame-hardening techniques, which involved oxy-acetylene-torching the surface of the plate. This was not only time-consuming, but it also used up a lot of gas. So much so that many ballistic trials were often done on RHA armor that hadn’t been surface hardened, and then math was applied after to calculate any additional effect provided by surface hardening – something that would prove disastrous later on.
To get this effect to work the steel itself would have a higher than normal carbon count, and by flame-treating the armor to a depth of 5-10% the metal would form a hard outer shell and soften towards the back to form a more ductile rear plate. This was very important, as by having a harder surface than the armour-piercing (AP) round, the round can be shattered. Then, the energy from any penetration that did occur could be dispersed through the steel by the softer inside to prevent perforation and spall.
Tank Armor
Generally, the harder a specific material is, the less toughness it has. Hardness refers to how much pressure must be applied to deform a material, while toughness refers to how much energy the material can absorb before it yields. Thus, the purpose of the softer steel under the hardened surface was actually to absorb the energy of an incoming round if it was successful in penetrating through the hardened surface.
This is why there are all of those pictures of armor with shells sticking out of it – the armor was sufficiently tough to absorb all of the kinetic energy of the shell without letting the shell pass through the plate
Of course, Germany was not the only nation to have been looking into this subject. The UK had long used face-hardened plate, but not heterogenous armor. They had discovered that post hardening, any weld seams were particularly weak, as carbon-rich metal in the 0.40 to 0.60% level does not like to bond via welding without a lot of treatment and cooling, and will still have stresses built into it that are likely to shear under any impact. To add to these woes, the welding itself applied a heat treatment in that area of the metal, further weakening it.

Thus nations like the UK initially used riveted armor. This involved taking a steel plate, cutting it to shape and predrilling it, then surface-hardening the metal by heating it in a mix of bone and charcoal. This would infuse the plate with added carbon, making it harder. The plate was predrilled and cut to shape beforehand, as if done afterward the metal was prone to shattering. This was then cooled and lined up with a corresponding softer framework of ductile material and the two parts riveted or bolted together.
Read More Char 2C – The Biggest Operational Tank Ever Built
This would help break up any AP shot when stuck horizontally, and the softer inner side would absorb shock and prevent heavy spalling. But it was not a perfect solution; firstly, it could lead to rivets or bolts breaking off inside the vehicle due to the stress on the two plates, although this was not as common as people like to believe. The second fact was that while such armor was more effective against solid tipped rounds, it was less effective against capped rounds.
Cast armor was more effective via the latter. The French realised this, so their tanks kept heavy cast plate in their designs for a lot longer.

The riveting method was however only effective to a certain point, after which the corresponding bolts did not scale in a linear way with the required increase in armor thickness, and so became impractical after a certain threshold was passed. Because of this, the UK switched to cast and welded steel.
There was also a further problem with the German method, particularly those steel plates that were to be used on Panther, and for that, we need to look at ships, warships in particular. During the First World War, a lot of ships had a mix of homogenous steel, with surface-hardened armor on the areas most likely to be struck by a broadside at perpendicular angles.
While early ships were designed to weather broadsides at relatively close range, as the range increased the curving arc of shots meant that they were striking the armor at greater angles. This was bad, as it was found that the more brittle face-hardened armor was inferior to RHA when hit at an angle, often allowing the rounds to pass through where regular RHA would have prevented this.
The Germans ironically were aware of this issue, with homogeneous Krupp-type armor coming into use for this very reason (the irony will make sense soon). So to surmise, the more angled the face-hardened plate was, with the aim to deflect shots, the more it proved incapable of doing so.
Now back to tanks.
The German rolled armor had an inherently high carbon content of between 0.26 and 0.53%, up to 0.75% in some samples, particularly in the middle range of plate thickness, along with high amounts of chrome added, up to 3.2%.
So, let’s look at the chemical composition of the plates used in the Panther, notably the 35-50 mm plates and the 55-80 mm plates.
Carbon: This is generally considered to be the most important alloying element in steel, and can be present up to 2%. However, anything over 0.50% will make welding extremely difficult and the metal brittle, particularly if it’s not been tempered. The reports from Panthers found in France showed levels of up to 0.63% to as high as 0.75%, which was deemed utterly unsuitable for steel designed to absorb impacts.
Sulfur: Usually an undesirable impurity in steel rather than an alloying element. In amounts exceeding 0.05%, it tends to cause brittleness and reduce weldability. In US examinations the Sulphur content in Panther armor was found to be 0.22%
Molybdenum: This element is a strong carbide former (carbides are materials that contain metal and carbon) and is usually present in alloy steels in amounts of less than 1%. It increases hardenability and elevated temperature strength and also reduced pitting and corrosion. The Germans began to run short of this material in early 1941, and by 1944 it was rare to find it in their tank armor. The results of this can be seen on surviving tanks with pitting on the metal surfaces.
Manganese: Steels usually contain at least 0.30% manganese because it assists in the deoxidation of the steel to prevent the formation of iron sulphide and inclusions, and promotes greater strength by increasing the hardenability of steel. However, if you have too much manganese and too much carbon, the steel becomes even more brittle. Both plates on the Panther have three times as much as required at between 1 to 1.2% Manganese.
Silicon: Usually only small amounts (0.20%) are present in rolled steel when it is used as a deoxidizer. Silicon dissolves in iron and tends to strengthen it. Weld metal usually contains approximately 0.50% silicon. German armor varies with slightly more or considerably less than required, between 0.32 and 0.80%
Chromium: This is a powerful alloying element in steel. It strongly increases the hardenability of steel, and markedly improves the corrosion resistance of alloys in oxidizing media. Its presence in some steels could cause excessive hardness and cracks near welds. The German armor samples varied between 1.25% and 2.13% or higher.
Nickel: Added to steels to increase hardenability. It often improves the toughness and ductility of the steel, even with the increased strength and hardness it brings. It is frequently used to improve toughness at low temperatures, but no amount was found on Panther tanks. It did however often appear in Tiger II steel.
Vanadium: The addition of vanadium will result in an increase in the hardenability of steel. It is very effective, so it is added in minute amounts. At greater than 0.05%, there may be a tendency for the steel to become embrittled during thermal stress relief treatments. The German examples were found to have between 0.14 and 0.30%
Phosphorus: Generally considered to be an undesirable impurity in steels. It’s normally found in amounts up to 0.04% in most carbon steels. In hardened steels, it may tend to cause embrittlement. German armor seemed okay here with 0.05%.
Making German Steel
So now we know the makeup of German steel, let’s explain the process.
First, we have to look at the steel-to-iron spectrum. The lower the carbon content the more ductile and softer the material. Steel typically contains less than 2% carbon. Once carbon gets to about 2%, it becomes iron, with cast iron at about 2-4%, pig iron at 6%, and cementite at 6% carbon.
Read More The Churchill AVRE Left Buried Under a D-Day Beach for 30 Years
For the Germans to get their armor to a heterogeneous level (the combination of flexible RHA, and a hard surface we mentioned earlier) you have to transform the iron crystals, or space lattice, to form a new shape. This is done via allotropy, by heating the metal over its threshold. The more carbon the lower the threshold, with the aim to get the steel somewhere between ferrite, at 0.02% carbon, and austenite, at 2% carbon.

As this transformation takes place the grain changes into a laminated structure of layered ferrite and cementite. To draw the carbon via diffusion to one side of the steel plate (for the hard face), it has to be heated and slowly air-cooled on one side; a time-consuming process, but one that will give your metal a harder surface and a more ductile inner side.
However, there is a shortcut: quenching the metal. This will rapidly cool the steel and won’t draw the carbon, but trap it in the steel, forming martensite (extremely high concentrations of carbon) in the steel, distorting the metal lattice. This makes the metal very hard, but also very brittle, which will make the metal prone to breaking and welding problematic. These two metals can be quickly examined by taking an angle grinder to a Panther and looking at the spark color.
So with the basic science bit out of the way, what did this mean for the Panther? Well quite a lot.
Why the Panther’s Armor was Poor
As we mentioned above, it was discovered that if the armor was treated correctly and gas heated and cooled, it formed a harder surface and softer ductile inner side (heterogeneous). However, we also mentioned that while this might have been effective for vertical armor, heterogeneous armor was less effective when sloped to deflect shots.
Unfortunately, the Panther’s armor was heterogeneous and sloped.
So this is our first problem; the Panther’s heterogeneous armor being stuck, especially by capped rounds, was not as effective as pure RHA would have been when angled back at 55 degrees (the slope of the Panther’s frontal plate).

But it gets even worse, as the metal was not treated correctly and contained high amounts of carbon. This steel had in several cases not been tempered by gas torching, and retained a high level of hardness but was also brittle. Several vehicles examined had not been heat-treated and cooled, while others examined by the Americans had been quenched with oil. It’s known oil baths were used on Stug and Jagdpanther hulls at Brandenburger Eiserwek.
While cast armor of a certain thickness will tolerate this tempering, thinner rolled steel will not, and forms large martensite deposits in the metal which means that under strain the metal will simply crack open and shatter.
The results from these impacts were catastrophic. As the shot travelled through the armor, the metal around the shot could not compress as quickly as ductile steel, and would split along the crystalline layers in the metal, often for several feet. The inner layer of the armor, even if not perforated, would also spall as the shockwaves hit the void and reflected back as these waves overlapped, forming a tension wave that could not disperse, causing it to escape by tearing a chunk of the armor out. This would ricochet around the hull, in a style not too dissimilar to HESH mechanics

So why did all this take place, were the Germans not aware? They had a history of good-quality metals.
Certainly, Albert Speer wanted to up production rates and many of the slower, fine-tuning processes were done away with, smoothing of exteriors and corners cut.
The war turning badly against them and the need to get more vehicles out quickly would have been a key aspect.
Read More The Myth of the Tiger II’s “Porsche” and “Henschel” Turrets
Was their internal sabotage? We know large numbers of slave workers were pushed through the steel plants – anyone with the know-how could have caused problems. Or was it simply miscommunication and industrial shortfalls? With wide-scale bombing, smashed plants, damaged infrastructure, and so on, were their originally-rigid processing guides just ignored?






